The research project FAIR CHARM aims to apply findings from basic scientific research to clinical applications with the aid of high-resolution, real-time imaging. The EU is funding the project with six million euro. The range of expertise is covered by an interdisciplinary consortium of six international universities and research institutions as well as three enterprises. Also involved is a team headed by Frauke Alves at the Max Planck Institute (MPI) for Multidisciplinary Sciences and the University Medical Center Göttingen (UMG).
Biomedical research has produced many innovations in recent decades that give hope for new approaches to treating diseases like cancer. And yet it is still a challenge to bring the findings from the laboratory to clinical application so that patients and those affected can benefit from them.
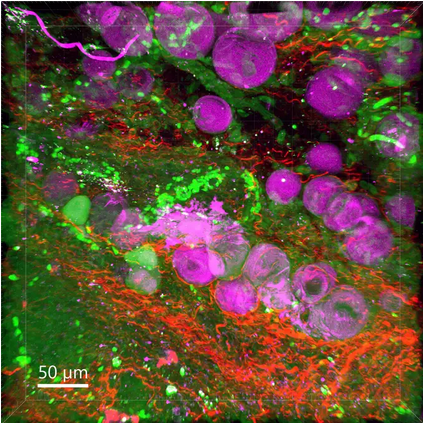
One of the reasons for this is the lack of imaging techniques that are able to map pathological changes both in individual components of a cell and in different cell types, even entire organs. New treatment strategies for different diseases therefore require special tools that allow to monitor multiple biological processes and make problems visible to researchers and practitioners.
“We aim to use new microscopy technologies to track biological processes involved in the onset and progression of diseases,” says Luigi Bonacina of the University of Geneva (Switzerland), who coordinates the FAIR CHARM (FAst Infrared Coherent HARmonic Microscopy) project. “The findings can then be used to develop new therapeutic concepts.” Among other things, the results should help to further develop the treatment of so-called muscular dystrophy with transplanted stem cells. Another possible field of application is the study of tumor and immune cells, how they interact and how they react to drugs. The project is divided into three main fields of action:
Three fields of action
The construction of the SWIM (Short-Wave Infrared Microscope), on one hand, will enable millimeter-deep, high-resolution imaging in tissue by using novel laser technology. With SWIM, researchers can penetrate tissue up to 15 times deeper than with conventional light microscopes.
The SLIDE microscope (Spectro-temporal Laser Imaging by Diffracted Excitation), on the other hand, promises to deliver unprecedented imaging speed of up to several thousand images per second, thus offering millisecond resolution for monitoring disease-relevant processes.
SWIM and SLIDE will be complemented by deep-learning recognition algorithms that will be adapted to the newly acquired observation data from the microscopes. The artificial intelligence is trained to recognize specific features for pathological changes in three-dimensional data sets, for example in the case of cancer and diseases of the brain or muscles.
The research group Translational Molecular Imaging headed by Frauke Alves at the Göttingen MPI for Multidisciplinary Sciences and the UMG focuses primarily on tracking tumor and immune cells as well as their interactions with the cells directly surrounding the tumor. In addition, the interdisciplinary team will look at mapping the transport, accumulation, and efficiency of new anticancer drugs in the body. “We will thus gather new information on the microarchitecture as well as on complex interactions between different cells and components of the extracellular matrix in tumor tissue. This way, we will investigate how these factors influence the efficacy of nanoparticle-based therapies,” Alves says. “This way, we want to harness the potential of basic research for clinical application and thus for patients.”
In addition to the Göttingen research group and scientists from the University of Geneva, multidisciplinary teams from Belgium, France, Hungary, Lithuania, and Germany are working together to optimize imaging methods and intelligent data processing with FAIR CHARM.
Contact
Prof. Dr. Frauke Alves
Research Group Leader at the MPI for Multidisciplinary Sciences and the UMG
+49 551 201-31655
falves@...